Stelara Biosimilar – Wezlana


Select Page
Posted by Michael Jannicelli | Nov 2, 2023 | Uncategorized | 0 |

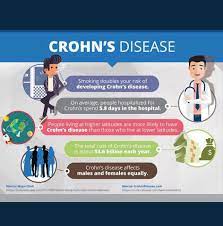
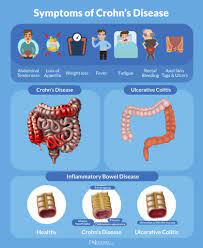
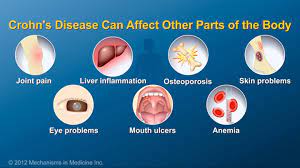
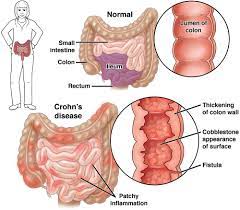
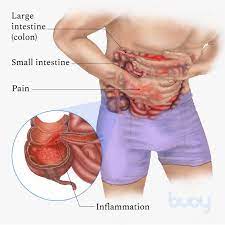
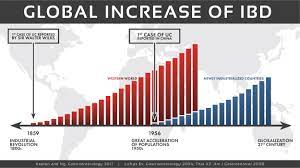
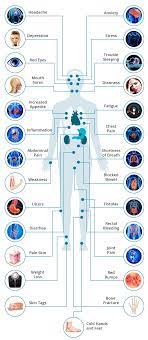
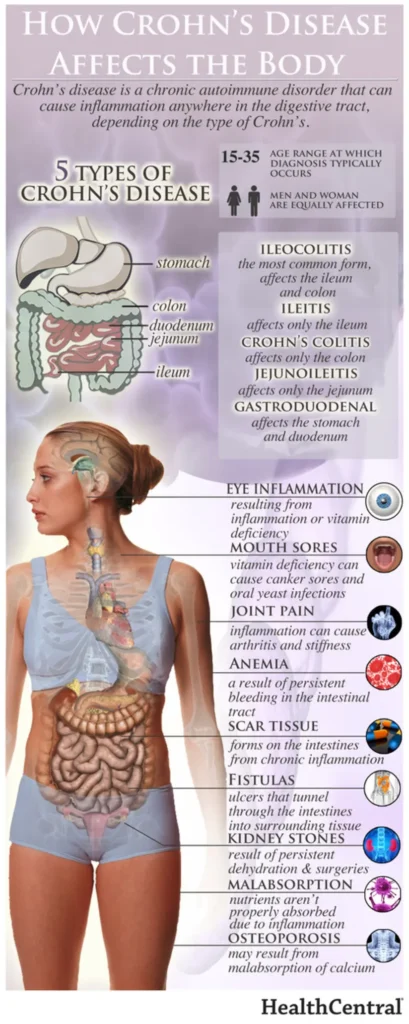
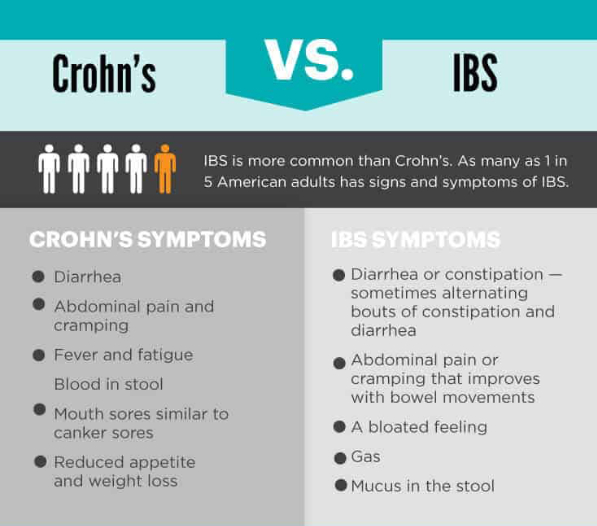
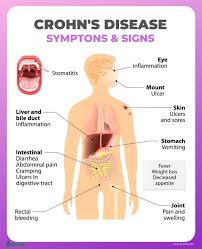
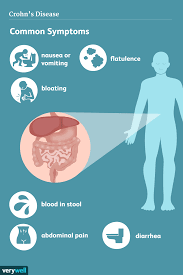
A biosimilar version of the multipurpose biologic drug ustekinumab has been approved by the FDA and can be swapped interchangeably with the original product sold as Stelara.The new version carries the brand name Wezlana and the generic name sports the suffix -auub, the FDA said in announcing the approval late…
Michael Jannicelli - "A PROVEN BUSINESS FIRESTARTER & BRANDING GENIUS" a true natural born Trendsetter. • Created over 100 products under 3 brands, sold in 30+ countries worldwide and influencing youth across the globe. • Featured in Forbes magazine +over 100 print publications, television talk shows, major motion pictures and national radio stations. • Co-founder and VP of Bliss beverage USA and President/co-founder of Socko International - fastest growing privately held beverage company in US (Forbes) with Hogan Energy & Raw Energy WWE. • Co-founder of Throwdown Industries (2003) - leading brand in multibillion dollar Impact/Action Sports Market (products designed for superior MN • Diagnosed with auto-immune diseases aged 13; using his experience to help others and promote a positive message while developing edgy products to benefit people with chronic illness.